Ferroelectric Materials
Calculations of Solid Properties Using Wien2k Package :: MATERIALS, TECHNOLGY AND APPLICATIONS :: INDUSTRIAL MATERIALS
Page 1 of 1
 Ferroelectric Materials
Ferroelectric Materials
Ferroelectrics are an important device in today’s world. They are useful both as capacitors, for example in camera flashes, or as non-volatile memory storage. The memory use of which you are most likely to be aware is in the Playstation 2.

To continue reading about the subject clik on the link below :
http://www.doitpoms.ac.uk/tlplib/ferroelectrics/index.php
 Re: Ferroelectric Materials
Re: Ferroelectric Materials
The ferroelectric effect was first observed by Valasek in 1921, in the Rochelle salt. This has molecular formula KNaC4H4O6·4H2O. The effect was then not considered for some time, and it wasn't until a few decades ago that they came into great use. Nowadays, ferroelectric materials are used widely, mainly in memory applications. This TLP will show how the ferroelectric effect arises, and how it is usefully used.
 Re: Ferroelectric Materials
Re: Ferroelectric Materials
Barium titanate
Let us consider one of the most well-known ferroelectrics, barium titanate, (BaTiO3).
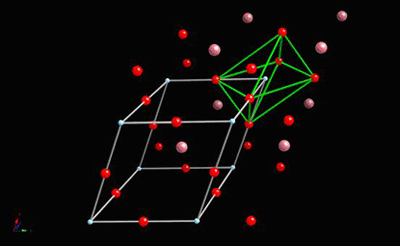
It has this perovskite structure:
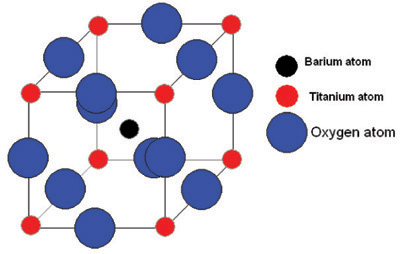

 Re: Ferroelectric Materials
Re: Ferroelectric Materials
Barium titanate and phase changes
The temperature at which the spontaneous polarisation disappears is called the Curie temperature, TC.
Above 120°C, barium titanate has a cubic structure. This means it is centro-symmetric and possesses no spontaneous dipole. With no dipole the material behaves like a simple dielectric, giving a linear polarisation. TC for barium titanate is 120°C.
Below 120°C, it changes to a tetragonal phase, with an accompanying movement of the atoms. The movement of Ti atoms inside the O6 octahedra may be considered to be significantly responsible for the dipole moment:
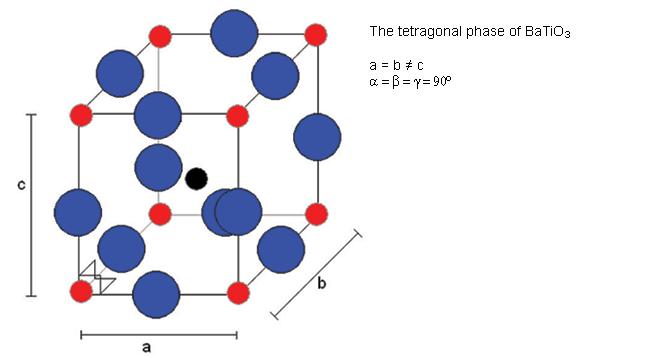
Cooling through 120°C causes the cubic phase of barium titanate to transform to a tetragonal phase with the lengthening of the c lattice parameter (and a corresponding reduction in a and b). The dipole moment may be considered to arise primarily due to the movement of Ti atoms with respect to the O atoms in the same plane, but the movement of the other O atoms (i.e. those O atoms above and below Ti atoms) and the Ba atoms is also relevant.
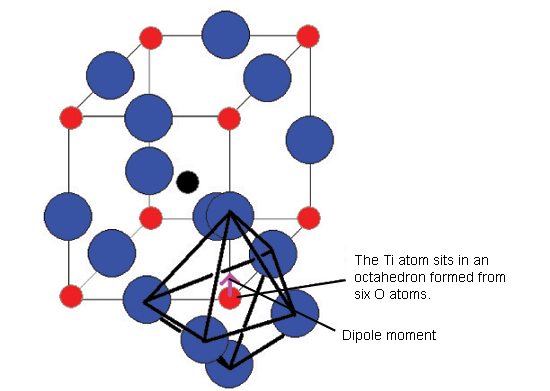
This shows the BaTiO3 structure with an O6 octahedron surrounding the important Ti atom.
The switching to a cubic structure is the reason for the polarisation spontaneously disappearing above 120°C. Barium titanate has two other phase transitions on cooling further, each of which enhances the dipole moment:
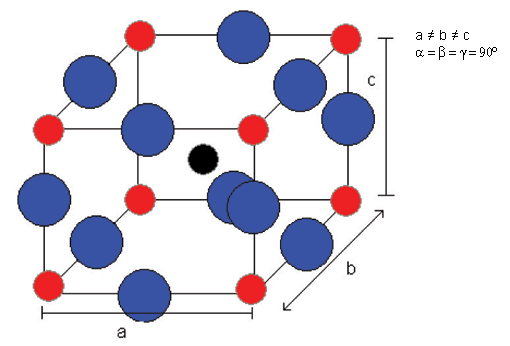
The phase which is reached after cooling to ~ 0°C from tetragonal is orthorhombic.
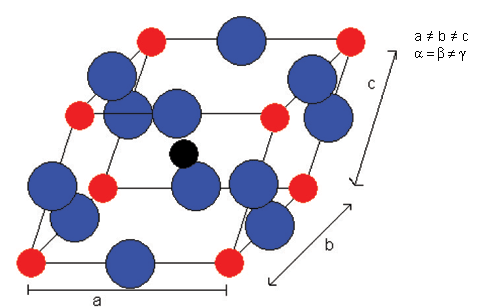
And then rhombohedral below -90°C:
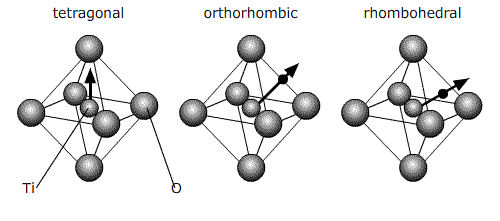
All of these ferroelectric phases have a spontaneous polarisation based to a significant extent on movement of the Ti atom in the O6 octahedra in the following way (using pseudo-cubic notation):
The temperature at which the spontaneous polarisation disappears is called the Curie temperature, TC.
Above 120°C, barium titanate has a cubic structure. This means it is centro-symmetric and possesses no spontaneous dipole. With no dipole the material behaves like a simple dielectric, giving a linear polarisation. TC for barium titanate is 120°C.
Below 120°C, it changes to a tetragonal phase, with an accompanying movement of the atoms. The movement of Ti atoms inside the O6 octahedra may be considered to be significantly responsible for the dipole moment:

Cooling through 120°C causes the cubic phase of barium titanate to transform to a tetragonal phase with the lengthening of the c lattice parameter (and a corresponding reduction in a and b). The dipole moment may be considered to arise primarily due to the movement of Ti atoms with respect to the O atoms in the same plane, but the movement of the other O atoms (i.e. those O atoms above and below Ti atoms) and the Ba atoms is also relevant.

This shows the BaTiO3 structure with an O6 octahedron surrounding the important Ti atom.

The switching to a cubic structure is the reason for the polarisation spontaneously disappearing above 120°C. Barium titanate has two other phase transitions on cooling further, each of which enhances the dipole moment:
The phase which is reached after cooling to ~ 0°C from tetragonal is orthorhombic.

And then rhombohedral below -90°C:


All of these ferroelectric phases have a spontaneous polarisation based to a significant extent on movement of the Ti atom in the O6 octahedra in the following way (using pseudo-cubic notation):

 Similar topics
Similar topics» Comparing the Weighted Density Approximation with the LDA and GGA for Ground State Properties of Ferroelectric Perovskites
» Cubic-tetragonal-orthorhombic-rhombohedral ferroelectric transitions in perovskite potassium niobate
» A tetragonal-to-monoclinic phase transition in a ferroelectric perovskite: the structure of PbZr0.52Ti0.48O3.
» Ferromagnetic and antiferromagnetic materials
» Different properties of materials
» Cubic-tetragonal-orthorhombic-rhombohedral ferroelectric transitions in perovskite potassium niobate
» A tetragonal-to-monoclinic phase transition in a ferroelectric perovskite: the structure of PbZr0.52Ti0.48O3.
» Ferromagnetic and antiferromagnetic materials
» Different properties of materials
Calculations of Solid Properties Using Wien2k Package :: MATERIALS, TECHNOLGY AND APPLICATIONS :: INDUSTRIAL MATERIALS
Page 1 of 1
Permissions in this forum:
You cannot reply to topics in this forum|
|
|

